A New Frontier for Health in Macau
Macau is making a serious play to become a major force in the life sciences. Driven by a strategic vision to diversify its economy and become a hub of innovation in the Greater Bay Area, the government is investing heavily in creating a world-class ecosystem for pharmaceuticals, biotechnology, and medical technology. This is an exciting time for the industry, but it’s also a field that is complex and highly regulated.
This guide, the core of our “Life Sciences” service hub, is designed to give you a clear and practical understanding of the legal landscape. We’ll walk you through the key regulatory bodies, the process for getting your products to market, and the unique opportunities that Macau offers, especially in the field of Traditional Chinese Medicine (TCM).
1. The Regulatory Maze: Navigating the Rules
The life sciences industry is not one where you can move fast and break things. The health and safety of the public is paramount, and the regulatory system is designed to ensure that all products are safe and effective. The main regulator you need to know in Macau is the Pharmaceutical Administration Bureau (ISAF). They are the gatekeepers, responsible for everything from clinical trials and product registration to manufacturing and marketing.
Navigating the ISAF’s requirements is a complex process that demands precision and expertise. The pharmaceutical law Macau is detailed and strict. Whether you’re seeking approval for a new drug or ensuring your facility meets Good Manufacturing Practice (GMP) standards, you need a legal partner who knows the system inside and out.
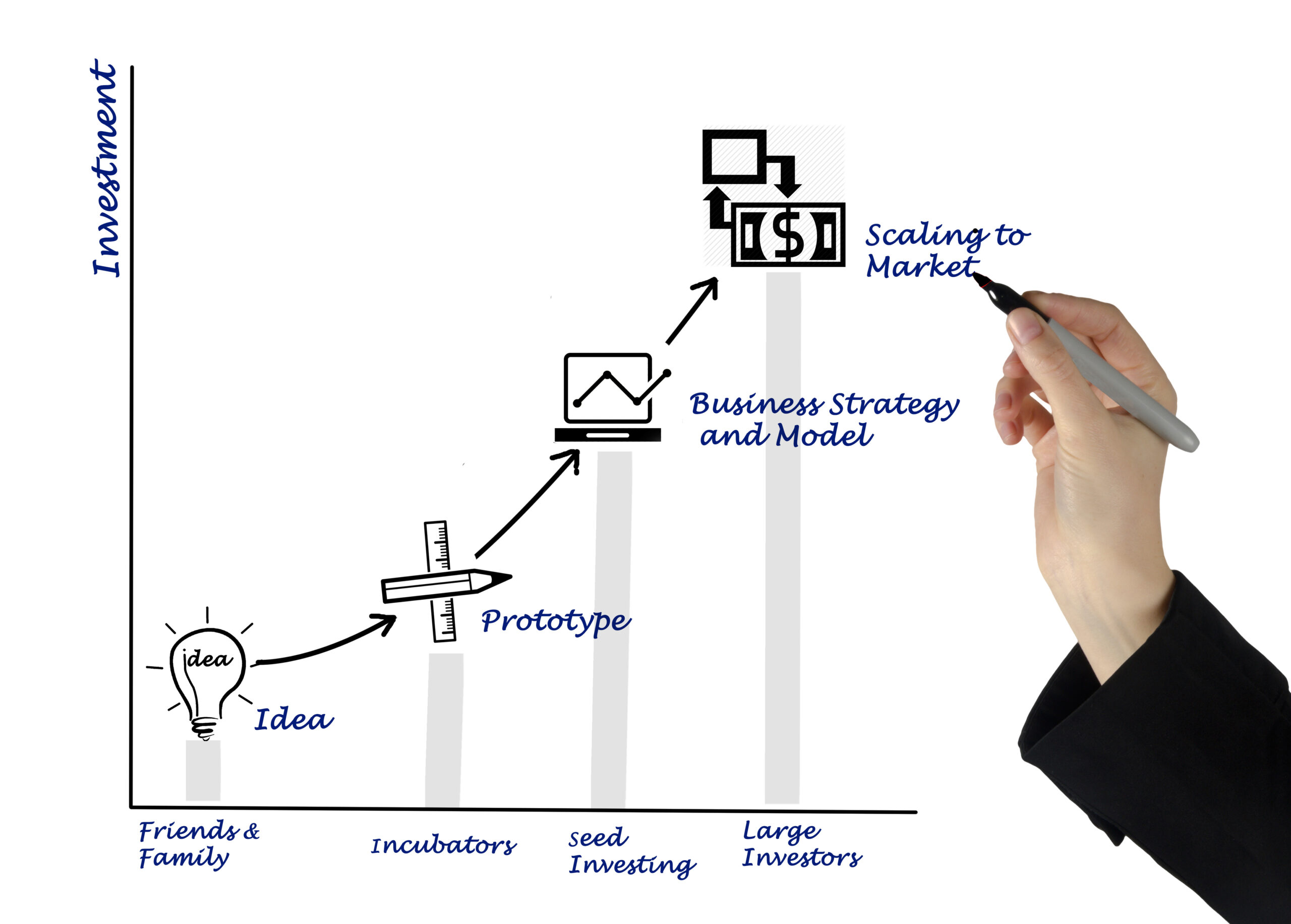
2. From Lab to Market: The Product Lifecycle
Getting a new drug or medical device to patients is a long and arduous journey. It starts with research and development, moves through rigorous clinical trials, and culminates in a detailed application for marketing authorization. Each step is governed by a specific set of rules.
Clinical trial compliance is a particularly critical area. Before you can test a new drug on people in Macau, you need to get approval from the authorities. This involves submitting a detailed protocol that demonstrates the scientific validity of the trial and the measures you have in place to protect patient safety. Once a product is on the market, your obligations don’t end. You have ongoing responsibilities for “pharmacovigilance” – monitoring the product’s safety and reporting any adverse reactions.

3. The Biotech Revolution: Fostering Innovation
Biotechnology is at the cutting edge of medical innovation, using living organisms and biological processes to create revolutionary new therapies. Macau is keen to attract and support these innovative companies. We provide specialized biotech legal services to help these pioneers navigate the legal challenges they face, from protecting their valuable intellectual property to structuring complex licensing and collaboration agreements.
4. The TCM Opportunity: A Bridge Between East and West
This is one of the most exciting aspects of Macau’s life sciences strategy. The government is pouring resources into the Traditional Chinese Medicine Science and Technology Industrial Park, a massive project in Hengqin. The goal is to create a global hub for the research, development, and standardization of TCM.
This creates a unique opportunity for companies in the TCM space. Macau is building a bridge between the rich traditions of Chinese medicine and the rigorous standards of modern international pharmaceuticals. For any company looking to be a player in the global TCM market, Macau is the place to be.




5. BN Lawyers: Your Partner in Life Sciences
Success in the life sciences sector requires more than just great science; it requires a deep understanding of the complex legal and regulatory environment. At BN Lawyers, we have a dedicated team that focuses on healthcare law Macau.
We provide comprehensive legal support to clients across the life sciences spectrum, from global pharmaceutical giants to innovative biotech startups. We can guide you through the entire product lifecycle, help you protect your intellectual property, and ensure you are fully compliant with all the relevant regulations, including the specific rules for medical device regulation. We speak your language, and we’re here to help you bring your innovations to the people who need them.
1. Who regulates the life sciences sector in Macau?
The main body you need to know is the Pharmaceutical Administration Bureau, or ISAF for short. They are the primary regulator for everything from drugs to medical devices.
2. Do I need government approval for a clinical trial in Macau?
Yes, you absolutely do. You can’t just start a clinical trial. You need to get the green light from the authorities, which involves submitting a very detailed application and protocol. They need to be sure the trial is scientifically valid and that patients are safe. This is a key part of clinical trial compliance.
3. How do I get a new drug approved for sale in Macau?
You have to go through a registration process with the ISAF. This means submitting a huge dossier of information about the drug’s quality, safety, and how well it works. If they are satisfied, they will grant you a marketing authorization.
4. What is Good Manufacturing Practice (GMP)?
GMP is a set of strict quality control standards for manufacturing pharmaceutical products. It’s all about ensuring that every batch of a drug is made to the same high standard. If you want to manufacture in Macau, you need a GMP license.
5. Can I advertise my prescription drug to the public?
No, that’s a big no-no here. Advertising prescription-only medicines directly to consumers is prohibited without specific authorization.
6. What is “pharmacovigilance”?
It sounds complicated, but it’s really about safety. It’s the process of monitoring the safety of a drug after it’s on the market and reporting any adverse reactions. If you sell a drug in Macau, you have a legal duty to do this.
7. How is patient data handled in Macau?
Very carefully. Patient health information is sensitive personal data, and it’s protected by Macau’s Personal Data Protection Act (PDPA). You need to have strong security measures in place to protect it.
8. What’s the deal with the Traditional Chinese Medicine (TCM) Industrial Park?
It’s a major government project located in Hengqin, right next to Macau. The goal is to create a world-class hub for the research, development, and global promotion of Traditional Chinese Medicine. It’s a big opportunity for companies in the TCM space.
9. Are the rules for TCM different from regular drugs?
Yes, they are. Macau has a whole separate regulatory framework for TCM products, covering everything from registration to manufacturing. It’s a specialized area of pharmaceutical law Macau.
10. How important are patents in the life sciences?
They are everything. A patent gives you a 20-year monopoly on your invention. That exclusivity is what allows companies to recoup the massive investment it takes to develop a new drug or medical device.
11. What is a “biotech” company?
A biotech company uses living organisms or biological systems to develop new products. In healthcare, this often means creating new drugs and therapies. We provide specialized biotech legal services for these innovative companies.
12. What counts as a “medical device”?
It’s a broad category. It can be anything from a simple tongue depressor to a complex MRI machine. The key is that it’s an instrument or apparatus used for a medical purpose. The medical device regulation is different from the rules for drugs.
13. Can a foreign life sciences company set up in Macau?
Yes, of course. A foreign company can set up a subsidiary or a branch here. You’ll just need to get the right licenses and approvals from the Health Bureau and other relevant authorities.
14. What are the opportunities in the Greater Bay Area (GBA)?
The GBA is a massive, integrated market. For a life sciences company, Macau can be a perfect launchpad to access that market, especially in areas like high-end healthcare services and TCM.
15. What is a CRO?
CRO stands for Contract Research Organization. These are companies that pharmaceutical and biotech firms hire to run their clinical trials and other research activities. They are a crucial part of the R&D ecosystem.
16. What is a “generic” drug?
A generic is a copy of a brand-name drug. It has the same active ingredient and works the same way. Generics can be sold after the patent on the original drug expires.
17. Is there any government support for R&D in Macau?
Yes, the government is keen to encourage innovation. There are various funding programs and tax incentives available for companies conducting research and development in Macau, including in the life sciences.
18. How do I import pharmaceutical products into Macau?
You need an import license from the ISAF for any products you bring in. The products themselves also need to be registered in Macau, and the whole process is subject to inspection.
19. How can BN Lawyers help my life sciences company?
We provide legal advice tailored for the life sciences industry. We can help you with the whole lifecycle of your product, from navigating the regulations for clinical trials and product registration, to protecting your patents and making sure you are compliant with the specific healthcare law Macau. We understand the science and the business, not just the law.
